UF Health researchers, collaborators get unprecedented view of gene therapy virus
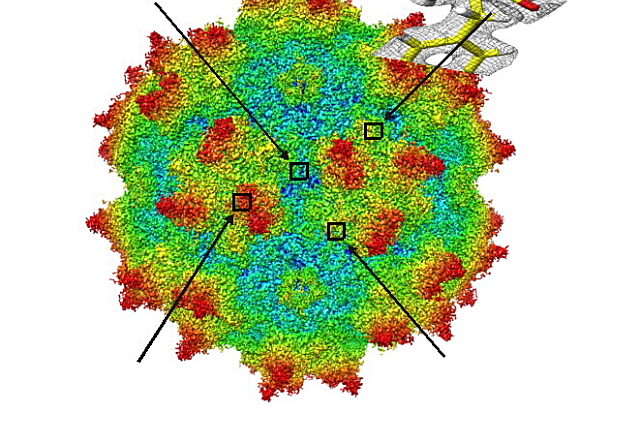
Researchers from University of Florida Health and the Salk Institute have obtained an unprecedented view of a gene-delivery virus, paving the way for further development of improved gene therapies.
Using an imaging method known as cryo-electron microscopy, researchers can see the shapes of biological molecules more closely than ever — almost down to the level of a single atom. That is crucial to further discoveries in gene therapy, which uses engineered viruses to deliver correctly functioning genes to a patient’s damaged cells.
The researchers created 3D, high-resolution images of a variant of adeno-associated virus serotype 2, or AAV2, with much better resolution than has ever before been accomplished. The findings were published Sept. 7 in the journal Nature Communications.
Seeing a virus more closely offers several advantages for gene therapy research. Among them: Scientists can gain a better understanding of how the human immune system recognizes viruses and how viruses enter the human body. It also provides an up-close view of the molecular mechanisms that make gene therapy effective.
Gene therapies are only effective if the engineered virus that delivers a new gene can evade the body’s normal immune response. This new way of seeing the AAV2 variant is the first step toward designing stealthier gene therapies that are effective for more people, said Mavis Agbandje-McKenna, Ph.D., a professor of biochemistry and molecular biology in the UF College of Medicine and director of the UF Center for Structural Biology.
“This technique will become especially important for developing a better understanding of how these viruses interact with the human immune system, which is one of the major remaining hurdles to the utilization of these viruses in gene therapy applications,” said Agbandje-McKenna, the paper’s co-senior author.
Gene therapy is being used or studied for a number of diseases caused by single-gene mutations, including Leber congenital amaurosis, which impairs vision; hemophilia, a blood-clotting disorder; and sickle-cell anemia, which distorts and kills red blood cells.
In the new study, UF Health researchers provided native and variant forms of AAV2 and collaborated with investigators at the La Jolla, California-based Salk Institute, who focused on a particular version of the AAV2 virus. The high-resolution information they obtained revealed a defect in the version of AAV2 they studied that was related to key changes in the region that the virus uses to transport its complete set of DNA.
“We applied a number of different procedures that have previously only been described in theory. We demonstrated experimentally, for the first time, that they can be used to dramatically improve the quality of this kind of imaging,” said Dmitry Lyumkis, Ph.D., a Salk assistant professor and structural biologist who was a co-senior author of the study.
Seeing the AAV2 variant in unprecedented detail and clarity also is important because it could allow researchers to understand the defect that was making it less effective, said Robert McKenna, Ph.D., a co-author of the study and a professor of biochemistry and molecular biology in the UF College of Medicine.
Mario Mietzsch, Ph.D., a postdoctoral fellow at the UF College of Medicine, helped to interpret the structure.
“Seeing is really believing,” he said.
The findings are the result of UF College of Medicine’s commitment to cryo-electron microscopy research that began several years ago, Agbandje-McKenna said. She is a co-founder of StrideBio, a North-Carolina based startup biotechnology company that focuses on developing AAV vectors that can evade neutralizing antibodies during gene therapy.
Researchers from the Columbia University, the University of North Carolina and the University of California – San Diego collaborated on the project, which was funded by the National Institutes of Health and Singapore’s Agency for Science, Technology and Research.
Related Videos
What is AAV and how does it work?
Research Landscapes: Mavis Agbandje-McKenna, Ph.D.
Mavis Agbandje-McKenna, Ph.D., researches the atomic structure of viruses as a way to develop gene therapies. She is a professor in the department of biochemistry and molecular biology at the University of Florida College of Medicine and a faculty member of the UF Genetics Institute.
About the author
