Putting a face to the name: Depression
Has a depression skeptic ever told you that it’s all in your head? Well, as a matter of fact, it is. Depression is a brain disease that takes a colorful world…

Update your location to show providers, locations, and services closest to you.
TMS is a safe and effective treatment for major depression and major depressive disorder.
Transcranial magnetic stimulation and therapy is an FDA-approved, noninvasive treatment for people with major depression who have not benefited from antidepressant medications. TMS and deep TMS are available by prescription only and are delivered under the supervision of a board-certified psychiatrist. TMS targets the key area in the brain that controls mood, the prefrontal cortex. This area is known to be underactive when someone is suffering from major depression. Using focused magnetic pulses to stimulate that area, TMS may restore normal function and reduce the symptoms of depression.Clinical trials have proved the effectiveness of TMS in treating patients with major depression. One out of two patients improved significantly, and one out of three patients was free of depression symptoms completely.
To schedule an evaluation, please call 352.265.4TMS (4687).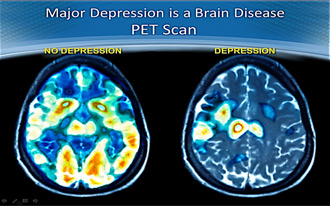 TMS treatments are free of side effects typically experienced with antidepressant medications.
TMS treatments are free of side effects typically experienced with antidepressant medications.





Has a depression skeptic ever told you that it’s all in your head? Well, as a matter of fact, it is. Depression is a brain disease that takes a colorful world…
